The 2008 movie The Curious Case of Benjamin Button [2], tells the story of a man who starts aging backward with bizarre consequences. Directed by David Fincher the protagonist of the movie was Brad Pitt. Is this phenomenon even possible in real life; apparently, many things that man thought was impossible are happening now. So this fantasy may someday become real.
Now coming to the scientific possibility of this fantasy. The fundamental question that we should answer is why do we age? The answer lies in our genes. Scientist has now discovered that the process of aging happens because of the shortening of a region in the chromosome called ‘Telomerase.’ Reversing aging involves lengthening of the telomeres that get damaged and shortened with the passage of time, a process that speeds up in the event of toxins entering the body — smoking, alcohol and air pollution are among elements that reduce the length of telomeres and therefore accelerating aging. Experimental studies done indicated that mice with hyper-long telomeres show less metabolic aging and longer lifespans [3]. The key to immortality may lie in our genes. One day if we can manipulate the telomerase shortening then the fantasy of immortality may be real.
Some other scientific research findings indicate that cancer cells become immortal once they undergo a transformation. HeLa is an immortal cell line used in scientific research. It is the oldest and most commonly used human cancer cell line. The line was derived from cervical cancer cells taken on February 8, 1951, from an African-American woman Henrietta Lacks, a patient who died of cancer on October 4, 1951. The cell line was found to be remarkably durable and prolific. HeLa cell line is the first immortalized cell line and one of the most iconic cell lines in medical research today [4].
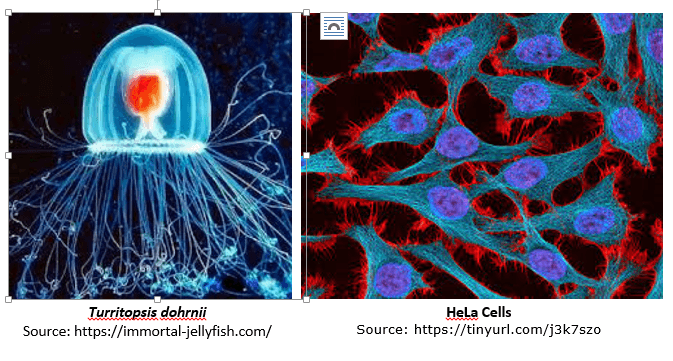
Scientists have also discovered a jellyfish that can live forever. Turritopsis dohrnii is now officially known as the only immortal creature. The secret to eternal life, as it turns out, is not just living a really, really long time. It’s all about maturity, or rather, the lack of it. Found in the Mediterranean Sea and in the waters of Japan, T. dohrnii is one of the few known cases of animals capable of reverting completely to a sexually immature, colonial stage after having reached sexual maturity as a solitary individual.
Like most other hydrozoans, T. dohrnii begin their life as tiny, free-swimming larvae known as planula. As a planula settles down, it gives rise to a colony of polyps that are attached to the sea-floor. All the polyps and jellyfish arising from a single planula are genetically identical clones. The polyps form into an extensively branched form, which is not commonly seen in most jellyfish. Jellyfish, also known as medusae, then bud off these polyps and continue their life in a free-swimming form, eventually becoming sexually mature. When sexually mature they have been known to prey on other jellyfish species at a rapid pace. If a T. dohrnii jellyfish is exposed to environmental stress or physical assault or is sick or old, it can revert to the polyp stage, forming a new polyp colony. It does this through the cell development process of trans differentiation, which alters the differentiated state of the cells and transforms them into new types of cells [5].
Along with telomerase attrition, there are many other factors responsible for the aging process such as genomic instability, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication. The current theories are assigned to the damage concept, whereby the accumulation of damage (such as DNA oxidation) may cause biological systems to fail, or to the Programmemed aging concept, whereby internal processes (such as DNA methylation) may also cause aging. Maybe someday we will be able to control these cellular processes according to our will with our advanced understanding of molecular & cellular biology. Once the causes of the aging is totally clear, immortality may be a really.
References:
1. Jinadu F. Can We Live Forever? Genetic Engineers say death will be optional by 2045. [https://tinyurl.com/qlvjwsp].
2. https://www.imdb.com/title/tt0421715/
3. Muñoz-Lorente M A, Cano-Martin A C, Blasco M A. Mice with hyper-long telomeres show less metabolic aging and longer lifespans. Nature Communications 2019; volume 10, Article number: 4723.
4. HeLa. https://en.wikipedia.org/wiki/HeLa
5. Richnov N. Can a Jellyfish unlock the secret of immortality? The New York Times Magazine Nov 28, 2012. [https://tinyurl.com/yx8xzru2]
Dr. Sanjoy Kumar Pal is a Professor of Biology in Skyline University Nigeria. He has a PhD. in Animal Genetics from Indian Veterinary Research Institute, India.
You can join the conversation on Facebook @SkylineUniversityNG and on Twitter @SkylineUNigeria
